*spoiler alert*
*duh*
Iron Man 2 is truly ridiculous. If anything, I barely know where to start with this movie. There is just so much dodgy science that it makes your head spin. In terms of characters, story line and you know, the stuff that’s kind of important to keep the audience interested and invested in its characters…. they kind of suck. Unfortunately the film succumbs to the ultimate failure of having pretty pictures and a lame story.
 If you don’t like it, you can just talk to the hand cause the face doesn’t want to hear it.
If you don’t like it, you can just talk to the hand cause the face doesn’t want to hear it.
But let‘s give credit where credit is due. Robert Downey Jr. is perfectly cast and he runs with what is given to him in style. His on-screen chemistry with Gwyneth Paltrow as Pepper Potts is genuine and enjoyable to watch. (Yes, I think the movie’s slow moments between the two of them work and it is entirely thanks to the actors that have to carry the lame script). And Mickey Rourke as a bad guy can do no wrong. But he is totally underutilized by not being given enough stuff to do. Or say. In his Russian accent. In short, the film is well acted and equally well cast, but very poorly written. Mostly it serves as an Avengers publicity stunt. You know, they made a movie to say they’re going to make another movie that will be much better. Thanks.
This brings me to the day I saw Iron Man 2 for the first time. I was watching it with my wife and a bunch of physics undergrads in a Marvel marathon. (My wife was a physics undergrad at the time). I was the only chemist in the bunch. We heckled the film during some of the most glaring contradictions and plot holes, all in good fun, but when the movie tried to pass sci-fi-science off in the form of distorted chemistry nomenclature, I fell off my rocker. Twice I had to pause and rant over how morosely incompetent the writing staff were. (The writing staff of the movie can’t all take the blame, most of these stem from flaws in the original comic book).
The first time was over ‘lithium dioxide’, which was injected into Tony Stark’s neck to combat the negative effects of the excruciating ‘palladium poisoning’ afflicting him due to his new heart/reactor (acquired from the first movie). To anyone who knows anything about chemistry nomenclature (it’s one of the first things you learn in chemistry class), you know that the combination of lithium (a metal) and oxygen (non-metal), will yield a product of their two most stable ionic forms. I.e. two lithium ions (both with a charge of +1) against an oxygen ion (with a charge of -2). The charges cancel out, see. Since the charges themselves reveal exactly how many atoms of which element are required to form a compound between the two, we do not need to specify how many atoms of which are present. The proper name would therefore be: lithium oxide.
But there are other forms of oxygen ions that can be formed. Specifically, if we wanted to form ‘lithium dioxide’, we could couple two oxygen atoms to a positively charged lithium ion. The two oxygen atoms can share one negative charge, but in that case it is called superoxide. The correct term would therefore be: lithium superoxide instead of lithium dioxide.
 Lithium superoxide even sounds much cooler than lithium dioxide! What gives?
Lithium superoxide even sounds much cooler than lithium dioxide! What gives?
It’s a little embarrassing how much this ticks me off. Mainly because it seems like you would have to go out of your way to intentionally get this wrong in order to produce the line ‘lithium dioxide’. It just doesn’t make sense.
The second time I had to stop the movie to rant was when Hammer was introducing some sort of ammunition that contained the compound “cyclotrimethylenetrinitramine”. This is actually the correct name of an explosive compound often designated as RDX. So, how can you get something that’s much more chemically complex right, but something like lithium dioxide so terribly wrong??
 Weapons grade high explosive. At least they got some things right.
Weapons grade high explosive. At least they got some things right.
This brings me to the scene that brought me to my knees in frustration and effectively paralyzed my brain. All it took for Iron Man to fix his synthetic heart and stop his continued exposure to Palladium poisoning was to “discover a new element”.
 Deeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeerrrrrrrrrrrrrrpppppppppppppppp.
Deeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeeerrrrrrrrrrrrrrpppppppppppppppp.
The periodic table was a way to organize all known elements according to their physical characteristics, reactivities, etc. It arranges all the elements according to their atomic number, which is the number of protons in the nucleus. It was astonishingly successful and even a few new elements were found as the original periodic table by Dmitri Mendeleev contained some “holes” where elements seemed to be missing. To make a new element you must “simply” add more protons to the nucleus and voila.
Recently, a slew of new elements were given names in honor of their discovery. These are elements 113-118 and the most stable of them have half-lives (time required for one half of the material to deteriorate) of just of few seconds. Meaning, they are incredibly unstable. Any other heavier elements that remain to be discovered are just as or even more so unstable than elements 113-118.
Or are they? If we give the writers the benefit of the doubt. The whole “Iron Man discovers a new element” jig, doesn’t have to be as mind-numbingly stupid as it originally seemed.
Islands of Stability
Glen Seaborg was a remarkable scientist. He is the only scientist to have an element named for him, whilst still alive. He pioneered the experiments which birthed superheavy elements, i.e. heavier elements than Uranium, nature’s heaviest naturally occurring element. As Seaborg started to bombard smaller elements into heavier ones (thus forming superheavy elements) he noticed a disparaging trend. Namely, as the masses of the newly formed elements increased, their stability and lifetimes decreased radically. He called this trend “the sea of instability”. As he saw it, as he formed heavier and heavier elements, he was walking up a peninsula of stable atomic nuclei. But the shore was approaching fast. Nothing stable remained ahead of him. Except for a theoretic combination of protons and neutrons in the nucleus.
Protons and neutrons are not in complete disarray within the atom’s nucleus. They are actually somewhat ordered in their configurative combination. As nuclear physics predicts, a very ordered and stable configuration of a precise number of protons and neutrons should form stable nuclei. More stable than existing for just a few seconds. Perhaps on the order of eons. This is what is called the island of stability.
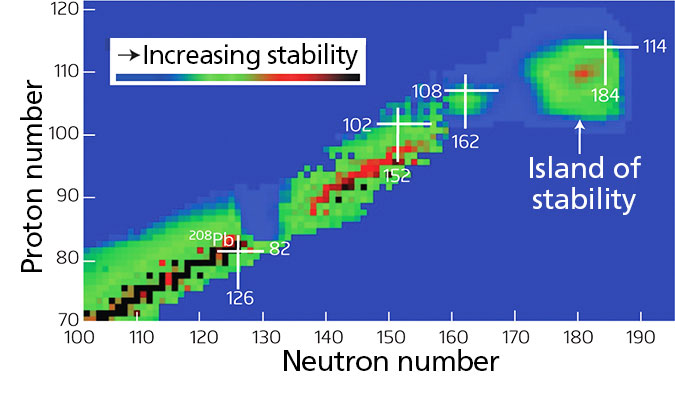 Was “Iron Dad” on to something? Maybe he really disliked Glen Seaborg not to entrust this information with him?
Was “Iron Dad” on to something? Maybe he really disliked Glen Seaborg not to entrust this information with him?
Is this what Tony Stark’s dad stumbled upon? Could the new element that saved Iron Man’s life just be among the nuclei found on the island of stability? Maybe. Exactly how Tony Stark synthesized the new element in the film does, unfortunately, make no goddamn sense whatsoever!
 Believe it or not, this is where they lost me.
Believe it or not, this is where they lost me.
To create these superheavy elements, heavier atoms must be accelerated to high speeds; fractions of the speed of light, before being bombarded onto heavier elements, in hopes that they combine to form an element that is a combination of the two atoms. In the film, Stark created a high-energy laser beam that he shot onto… some material… and thusly created the element whose inner architecture his dad hid in a model of a park. Maybe the light beam included some metallic atoms? Maybe it included quarts clusters accelerated to high speeds that all combined to reach the island of stability? Maybe the effective temperature of the radiation was high enough to induce some sort of a “local thermal atomic merger”.
Maybe it’s just all bullshit and the movie sucks. I mean, despite everything, the movie did catalyze me to familiarize myself a bit more with nuclear physics/chemistry. Did Citizen Kane manage that feat?

Saw a medical doctor break this down, and this post came up on a search. I’ve speculated that PdCl could form in Tony’s blood, which is insoluble, however it’s *possible* for it to be chelated with LiCl to form Li2PdCl4 which is soluble and can be urinated out. But Lithium Chloride doesn’t sound as cool.
LikeLike