Time has a way of getting away from us. This particular piece is one which I have been meaning to write for a while. Although I finished my Marie Curie fellowship officially in August 2021, I haven’t felt like I’ve been in a position to write this piece until now.
Back in 2019 I won a distinguished Marie Curie fellowship to go to the Leiden Observatory in the Netherlands to carry out a project on polycyclic aromatic hydrocarbons (PAHs) in the context of Astrochemistry. For those who don’t know, the Leiden Observatory is one of the most distinguished institutions in the world when it comes to astronomy and its alumni of directors include figures such as Jan Oort, Willem de Sitter, Ejnar Hertzsprung and Frederik Kaiser. Leiden is also a veritable hub for astrochemistry worldwide.
I wrote about my plans shortly after starting my fellowship and I used my website to upload updates on my progress during the project.
I had a meticulous plan laid out upon my start. The plan involved writing proposals, experimental work, beamtimes, etc. Six months after my fellowship started, the Covid-19 pandemic completely derailed my plans. It’s funny because in my original Marie Curie application, I had made detailed back-up plans in case some things didn’t work out; or a so-called risk register. What if the experiment completely malfunctioned? What if I didn’t get the beamtime I applied for? What if this? What if that?

No one could have predicted that the question: What if a global pandemic hits and immediately halts all international travel and forbids me going into the laboratory to perform experiments? Then what???
That was the issue I was faced with six months after starting my fellowship.
And let’s get one thing straight. It’s not like the fellowship started smoothly! To begin with it appeared to be very optimistic. In my first week I wrote two synchrotron beamtime proposals, one to go to SOLEIL and one to go to the SLS. February 2020 I was doing experiments at the SLS when news was coming out that something big was going on. A storm was on the horizon. I didn’t take it very seriously then because I tend not to take news very seriously in the grand scheme of things. I guess that is just my sense of feeling entirely desensitized to a constant onslaught of news about shootings, violence, crime, and politicians too corrupt to be allowed even a smidgeon of the power they possess.
But I digress.
A couple of weeks later, I was actually on tour with my former band, Misþyrming when everything started shutting down. Each city we played in appeared to go into complete shutdown the second we played our last note of the night. When it came to our final show of the tour in Tallinn, Estonia, we had finally gotten the news. Everything is closed and cancelled. The organizers actually managed to still put us up in a rehearsal space equipped with an amazing sound system, and multiple cameras which allowed us to stream the show. In the end 20000 people watched our live show on social media and youtube which was quite a larger audience than we had anticipated from the 200 tickets sold for the show in Tallinn.

Again, I digress.
Before going on that tour, I had been struggling with my experimental apparatus, the instrument for photodynamics of PAHs (or i-PoP, see below). This experiment was the first step in my ambitious plans for my Marie Curie fellowship. When it works, PAHs are sublimated in a small oven and the gaseous PAHs are ionized with an electron gun. The ionized PAHs, or PAH cations, are transported into a quadrupole ion trap where the ions are compressed into an ion cloud thanks to the quadrupoles and a He buffer gas. The contents of the trap are then irradiated with a laser and the resulting fragments are analyzed with a mass spectrometer, so experiment spits out series of mass spectra. There are a myriad of different things that need to be tweaked and/or kept stable so that the results can be compared to understand the influence of increased laser exposure, increased laser power, the laser wavelength, etc.
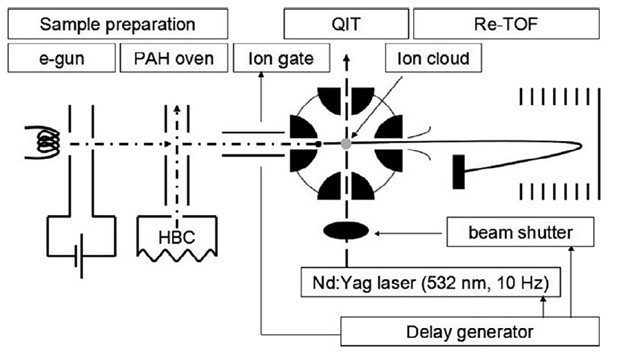
The SOLEIL proposal I wrote in my first week that got accepted included shipping the i-POP instrument from Leiden to SOLEIL and use the synchrotron light source to do experiments instead of the laser. However, the issues I was facing were that the oven wasn’t working as it should. I had been trying to calibrate the oven for quite some time before realizing that the heating element was busted. This happens a few weeks before the beamtime which was going to take place a week after I got back from my Misþyrming tour.
Ordering a new heating element would take too much time as it would be shipped to the Netherlands from California so I needed to do something different. Speaking with the researchers at SOLEIL (my former bosses), we came up with something different. I could use an on-site ion trap at SOLEIL to do similar experiments as I would have carried out on i-POP, but on significantly larger PAHs.
But then Covid-19 put everything on stand-by. No trip to SOLEIL. No going into the office to fix i-POP. So… what then?
Well. Talking with my office mates, it seemed that everybody at the observatory were exceptionally good at coding. And using Python. I figured, well, why don’t I use the time when I’m stuck at home away from the lab to learn how to code in Python? I took some online courses that had been made free as the pandemic hit which was very helpful.
Eventually, things started easing up and I was allowed back into the laboratory bit by agonizing bit. The laboratory at the Leiden Observatory hosts ten different experiments. However, only five people were allowed in the laboratory at a time. This made things very challenging and as a result I started working in excruciatingly long shifts, similarly to what I had done at the SOLEIL synchrotron during my 2017-2019 postdoc. However, these lab shifts were not followed with obligatory rest days as was the case at SOLEIL where, as a French civil servant, my own personal health was prioritized. I was tired. But I did a lot of good work which has culminated in two publications in the International Journal of Mass Spectrometry that I am very happy with (see here and here).
My research question was twofold. (i) How does the starting shape and symmetry of the PAH cations influence how they break apart and the fragments that are formed? (ii) How does the starting size of the PAH cations influence how they break apart and the fragments that are formed?
In the first publication I compare the fragmentations of three dibenzopyrene isomers. They all have the same starting molecular formula, C24H14, but their structures are different (see below).

Interestingly, they all seem to favor the formation of so-called “magic-number” carbon clusters. Specifically carbon atoms containing 11 to 15 carbon atoms. This is quite neat, I remember becoming very jubilant and excited when I saw that I could replicate these results. They weren’t a fluke.
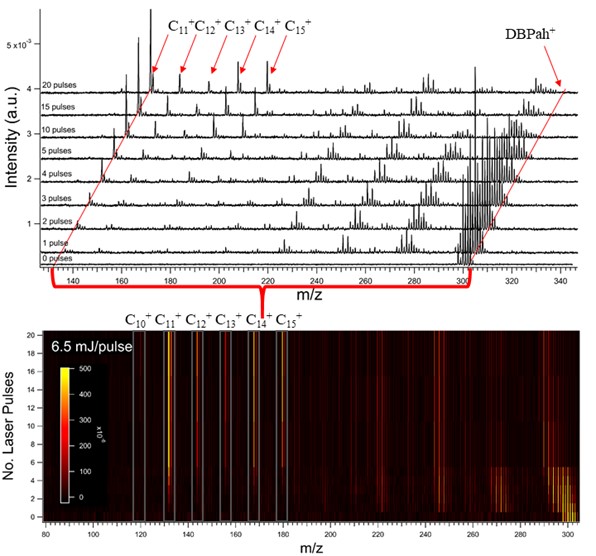
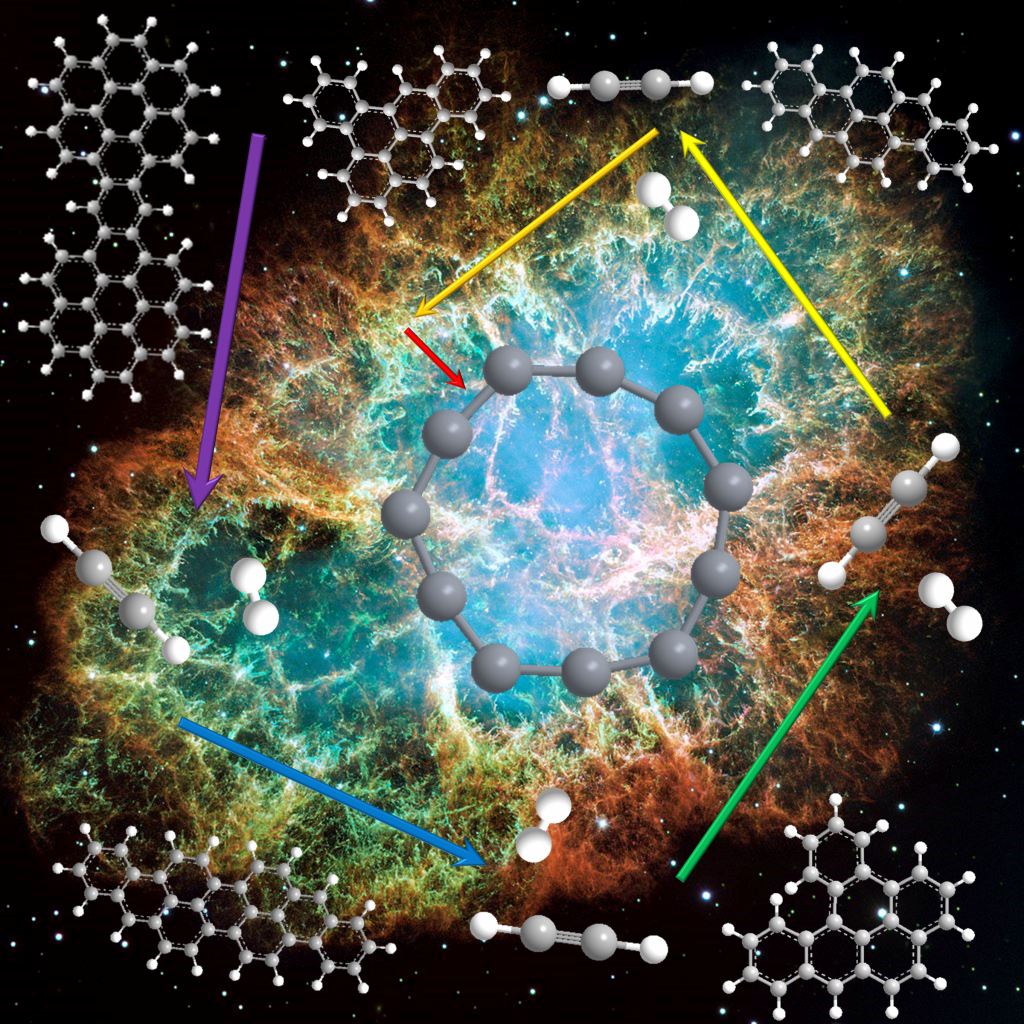
But what if we increase the size of the PAH cations? To answer this question, I investigated isoviolanthrene and dicoronylene (C34H18 & C48H20, respectively).
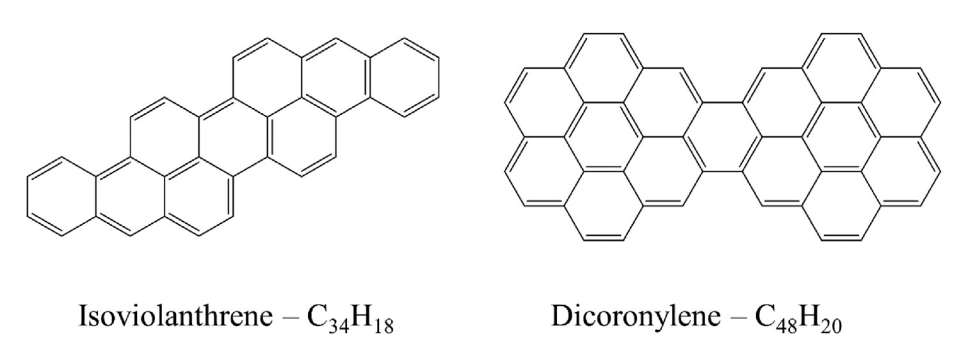
Shockingly, they exhibited the same magic-number carbon clusters, and in very similar branching ratios as the dibenzopyrenes! Furthermore, these carbon clusters are formed very early on in the dissociation process. Classically it has been shown that when PAHs break apart, they tend to favor losses of H atoms, H2 molecules and C2H2 or C2 molecular moieties. Here, it seemed that these carbon clusters were being formed in direct competition with the C2H2 & H/H2 losses. This is quite interesting because if all PAHs, regardless of size and symmetry, favor similar fragmentation products (these carbon clusters), then surely, they could be found in the same regions in space where PAHs are formed. Which is basically everywhere.
But then the question is, what is the structure of these carbon clusters?
The study of carbon clusters goes back to the 1990’s (see figure below from von Helden et al. 1991) where there was an enormous boom in carbon cluster research because of the discovery of the C60 fullerene molecule. Much work was devoted to carbon clusters, and we know now that the preferred structure of carbon clusters depends on the number of carbon atoms. What is the structure of the magic-number carbon clusters observed in my work on i-POP?
It’s rings. Carbon rings. So-called cyclo[n]carbons, where n denotes the number of carbons in the ring. If the clusters would be smaller, their preferred geometry is linear chains. If they are a bit larger, it would be butterfly or doubly ringed structures. If there are thirty C atoms or more, the preferred geometry are buckyballs.

So, we end up with carbon rings when we start with PAHs? How?
This is a hard question. I image that when PAHs absorb photon energy, the dissociation process is not very clear-cut. It is more dynamic and chaotic than a simple fragment being broken off and then rest of the energy radiated away through fluorescence or intramolecular vibrational redistribution (IVR). I believe that what we saw in the experiment is pointing towards a multiplexed and incredibly complicated potential energy diagram of a big molecule where aromatic bond ruptures are fairly frequent. This would open up the PAH carbon skeleton and quite easily allow the formation of carbon rings. To verify this hypothesis will require an enormous amount of quantum chemical computations, however. Hopefully, some might be interested in this project.
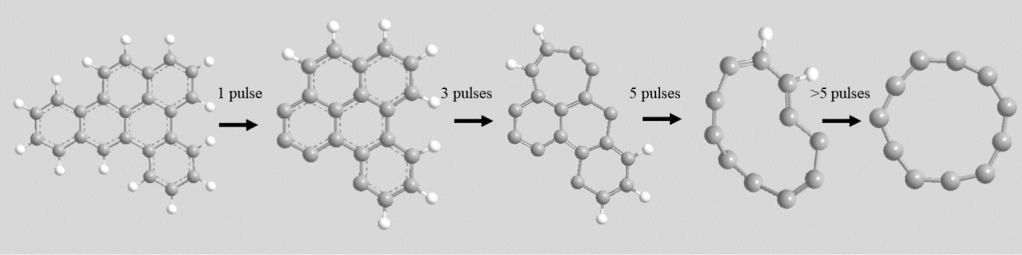
But if these carbon rings are so easily formed from PAHs, how would we go about detecting them in space?
This might even be a harder question than the previous one. These carbon rings, particularly when they lose electrons, undergo various geometrical, so-called Jahn-Teller, distortions. These are incredibly difficult to calculate which makes it a very challenging quantum chemical task to verify their geometry and compute their vibrational signatures. But it is also super challenging to create them in the laboratory in the first place under controlled conditions such that their spectra and dynamics could be investigated.

It sort of feels like that with my Marie Curie fellowship I stumbled upon a really interesting cosmic riddle, but because academia only employs early-career researchers for a couple of years at a time, I can’t take this much further for the time being.
To me, this is still super interesting, and I really hope that others think so as well so that they can maybe answer some of the questions my work sparked.
So this was a short story about the first part of my Marie Curie fellowship. When I get my other projects published, I will write about them in more detail.

One thought on “My Marie Curie Individual Fellowship (2019 – 2021(2)) – Part 1 of X, X = 2-5”